jward
passin' thru
newsweek.com
Can Blood from Young People Slow Aging? Silicon Valley Has Bet Billions It Will
Adam Piore
28-35 minutes
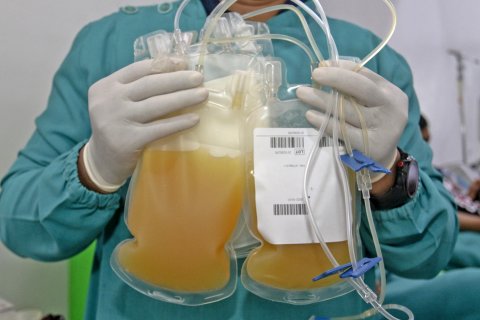
The Spanish firm Grifols set off a kerfuffle last year when it nearly doubled the going price for blood donations for a COVID-19 treatment trial. Brigham Young University in Idaho had to threaten some enterprising students with suspension to keep them from intentionally trying to contract COVID-19. The trial failed, however, and now the Barcelona-based firm is hoping to extract something far more valuable from the plasma of young volunteers: a set of microscopic molecules that could reverse the process of aging itself.
Earlier this year, Grifols closed on a $146 million-deal to buy Alkahest, a company founded by Stanford University neurologist Tony Wyss-Coray, who, along with Saul Villeda, revealed in scientific papers published in 2011 and 2014 that the blood from young mice had seemingly miraculous restorative effects on the brains of elderly mice. The discovery adds to a hot area of inquiry called geroscience that focuses on identifying beneficial elements of blood that dissipate as we age and others that accumulate and cause damage. In the last six years, Alkahest has identified more than 8,000 proteins in the blood that show potential promise as therapies. Its efforts and those of Grifols have resulted in at least six phase 2 trials completed or underway to treat a wide range of age-related diseases, including Alzheimer's and Parkinson's.
Alkahest and a growing number of other geroscience health startups signal a change in thinking about some of the most intractable diseases facing humankind. Rather than focusing solely on the etiology of individual diseases like heart disease, cancer, Alzheimer's and arthritis—or, for that matter, COVID-19—geroscientists are trying to understand how these diseases relate to the single largest risk factor of all: human aging. Their goal is to hack the process of aging itself and, in the process, delay or stave off the onset of many of the diseases most associated with growing old.
The idea that aging and illness go hand and hand is, of course, nothing new. What's new is the newfound confidence of scientists that "aging" can be measured, reverse-engineered and controlled.
Until recently, "people working on diseases did not think that aging was modifiable," says Felipe Sierra, who recently retired as director of the Division of Aging Biology, part of the National Institutes of Health. "That is actually what many medical books say: The main risk factor for cardiovascular disease is aging, but we cannot change aging so let's talk about cholesterol and obesity. For Alzheimer's, aging is the main risk factor—but let's talk about the buildup in the brain of beta-amyloid proteins. Now that is beginning to change."

Felipe Sierra, who recently retired as director of the Division of Aging Biology, part of the National Institutes of Health. Courtesy of NIH/National Institute on Aging
The result is a flood of investment money, an explosion of research into what precisely goes wrong in our bodies as we get old and the promise of clinical results down the road.
In the months before the pandemic, investors ponied up billions of dollars to fund biotechs aimed at commercializing the new science. Some biotech firms are developing drugs and infusions designed to clean up zombie-like cells and metabolic junk that accumulate with age. Others hope to infuse new vigor into flagging cellular components, such as stem cells, or spur the body into beneficial actions by adding obscure hormones or proteins, that decrease as we get older. The NIA, under its director, Richard Hodes, recently announced plans to spend about $100 million over the next five years on basic research aimed at understanding "cellular senescence."
"You have no idea how many people are interested to investing money in longevity," said Nir Barzilai, the founding director of the Institute for Aging Research at Albert Einstein College of Medicine of Yeshiva University, and the founder of a company aimed at mitochondrial health. "There are billions of dollars."
Although the vast majority of these efforts remain in preclinical development, several have recently entered FDA trials and could potentially hit the market in a few years. Some are already appearing on the gray market, raising concerns that hucksters are peddling anti-aging snake oil. Others, meanwhile, worry what might happen if these drugs actually do deliver on their promise: Will poor young people be coerced into selling their blood to elderly billionaires? Will magical anti-aging pills become the province of the Park Avenue and Hollywood rich, like facelifts, hair plugs and botox injections? Will the rest of us senile peasants be forced to watch them age backwards as we are left to wither and die?
Hacking Old Age
Getting old usually doesn't end well. Despite massive gains in human life expectancy—over the last 150 years, it has almost doubled in many developed nations, though it dipped in the past year due to the pandemic—we still haven't found a way to halt the relentless toll time takes on our bodies.

Older people practicing yoga at an assisted living facility to help stay limber; Silicon Valley is working on its own solution to the mobility issues and other problems of older age. Craig F. Walker/The Boston Globe/Getty
When we grow old, our immune systems begin to break down, creating a state of low-level inflammation that suppresses cellular regeneration and leads to those aches and pains. We may be living longer, but we are hurting. As our mitochondria cease to effectively produce energy for our cells, we spend many of those hard-won extra years taking afternoon naps. As our stem cells grow lethargic and quiescent, our muscle mass shrinks and our bones become increasingly fragile. In short, our bodies fall apart.
The first hint that hacking the biology of aging might be possible came, improbably enough, from a series of laboratory experiments on a lowly species of roundworm. In the late 1980s and early 1990s, studies of identical twins had already shown that about 30 percent of longevity in humans could be attributed to genetics. But most scientists believed the process of aging was far too complex a phenomenon to modulate simply by tweaking a couple genes or taking a pill.
Then in 1993, Cynthia Kenyon, a biologist at the University of California in San Francisco, doubled the lifespan of a worm from three weeks to six by mutating a single gene. The work suggested the possibility of drugs that could increase longevity without targeting specific diseases. It suggested that the process of aging itself might be manipulated.
At the time, a fringe group of longevity enthusiasts were, unknowingly, already trying to do what Kenyon did in roundworms through the practice of calorie restriction. This idea took off when gerontologist Roy Walford of UC Los Angeles doubled the lifespan of lab mice by restricting their caloric intake and then became a vocal advocate for the practice in humans. He penned a series of best-selling books in the 1980s and adopted a 1,600-calorie-a-day diet for the last 30 years of his life. Today, his emaciated acolytes can still be spotted at longevity conferences, meticulously measuring out their food during lunch breaks.
Emerging science now explains why there may be something to this method of bio-hacking. In humans, insulin is the hormonal signal that cues our cells to absorb sugar and convert it to energy. Along with a closely related hormone called Insulin like-Growth Factor 1 (IGF1), insulin affects a large number of other cellular processes, including the rate of cellular division, which many believe is directly related to aging. When the insulin and IGF1 in humans or analogous compounds in worms are dialed down—because we are starving, or the genes have been tweaked—a host of cellular repair mechanisms that are normally on standby kick into high-gear.
The adaptation makes sense from the perspective of evolution. Prehistoric times consisted of long periods of scarcity punctuated by precious windows of abundance. Since there was no telling how long the good times would last, our ancestors evolved the ability to grow and build fat and muscle as quickly as possible. A feast triggered the body to release insulin and IGF1, which allowed our cells to begin absorbing glucose and spurred them to pour energy into cellular reproduction and regeneration for as long as our insulin levels remain high.
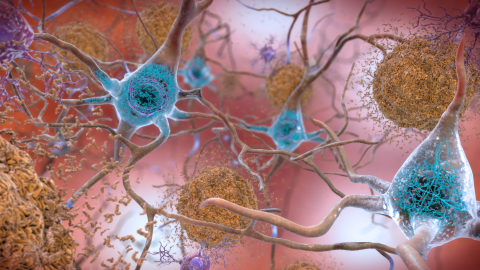
Beta-amyloid plaques and tau in the brain. Courtesy of NIH/National Institute on Aging
When calories were no longer easy to come by, our bodies adjusted by lowering insulin and IGF1 levels—a cue for our cells to slow regeneration and reproduction, and instead divert energy into cellular processes most likely to facilitate our survival through cold, lean times. The human body protects cells it already has: it produces more enzymes to ensure proteins don't misfold, it ramps up the machinery designed to repair broken DNA and it breaks down cellular debris and defective cells it might otherwise ignore, scavenging for parts it can use to feed its healthier cells through the lean days. In the process, it cleans up cellular garbage that, particularly as we grow older, likely promotes low-level inflammation.
Currently there is no FDA-approved indication for drugs that target the process of aging itself. To win approval, drugs must target a specific disease. It's no coincidence that some gerontologists have chosen the popular diabetes drug metformin to serve as the "template" for a new class of FDA-approved anti-aging drugs. It works by influencing the body's sensitivity to insulin and can have an effect on the pace of metabolism and energy expenditure. If studies now underway confirm its efficacy and it wins approval, it would create an entirely new regulatory category of drugs that, rather than treating specific diseases, will work to prevent our bodies from deteriorating further once we have already developed one.
"Aging drives disease," says Albert Einstein's Barzilai, who is leading the study. "That's our point. If we stop aging, it stops driving the diseases."
A whole host of other potential anti-aging-drugs might not be far behind. Molecular biologists discovered a second important way to manipulate the aging process in the early 2000s by breeding strains of baker's yeast, fruit flies and worms to live longer. They sequenced the genomes of the hardiest strains and then worked backward to identify the specific genetic variants that seemed be associated with increased longevity. That led them to mTOR, a genetic variant that serves as the target of an anti-aging drug called rapamycin, which is now undergoing clinical trials. Among these scientific efforts is a massive longitudinal study of canine aging at the University of Washington, which has enrolled 30,000 dogs and plans to select 500 lucky pooches to participate in a rapamycin trial.
The hunt for other "control knobs" that might be targeted with drugs continues at research institutions around the world. A third approach would increase the production an enzyme called AMP-Kinase, which modulates cellular processes like growth and metabolism, based on the level of energy available for consumption. It, too, has emerged in recent years as another promising target, along with many other molecules, hormones and proteins that appear to decline with age and that play a key role in cellular repair, regeneration, protection and efficient function.
Vera Gorbunova, co-director of the Rochester Aging Research Center at the University of Rochester, and her colleagues compared 18 rodent species, including, beavers, hamsters and mice, sorted them by longevity, and looked for interesting patterns. Significantly, they found that "more robust" repair of broken DNA, the molecular-level cellular blueprints found in every one of our cells, seems to coevolve with longevity. In other words, species that live longer have a more robust ability to fix the kind of problems that inevitably occur with age. The research was recently published in the journal Cell.

Vera Gorbunova, co-director of the Rochester Aging Research Center at the University of Rochester University of Rochester
"There was a very strong correlation to how long different species live and how well they repaired DNA breaks," Gorbunova said. "We also look at why it's better. We found a protein that is very important in DNA repair and it is more active in long-lived species."
Gorbunova has done some of her most exciting work on an obscure superstar in the longevity field known as the naked mole rat, a furless, wrinkled rodent with a pair of beaver-like incisors that lives in tunnels in East Africa. The rat, sometimes referred to by its cuddly nickname, "Sand Puppy," has an unusually long-life expectancy—30 years, 10 times longer than its close cousin, the North American field mouse, which rarely makes it past three.
Gorbunova traces much of the hardiness of the mole rats to an abundance of hyaluronic acid, a major component of skin that is involved in tissue regeneration. Although mice and humans also have hyaluronic acid, the tissues of naked mole rats are "saturated with it," says Gorbunova. In addition to having strong antioxidant properties, and others that seem to attenuate the destructive consequences of the chronic, widespread inflammation that often accumulates with age, the abundance of hyaluronan also seems to prevent the growth of malignant cancer cells.
"Hyaluronan is a very nice story because we can see the possibility of translating it to humans," Gorbunova says. "We have it, but we don't have a lot of it, so I think there is room for improvement. We can find ways to increase our own levels of hyaluronan."
To Gorbunova, the differences between the mouse and naked mole rat are easily explained by evolution—their respective adaptations are geared toward increasing their chances of reproductive success. "For a mouse, the best strategy to have more progeny is to be very, very prolific very quickly because then somebody's going to eat it, and it just doesn't have a chance to live longer," she explains. "The naked mole rat lives underground and has very few predators. And they breed until very late in life. So they would evolve the mechanism to allow them to live longer and to breathe as long as possible just because they can. No one is there to eat them. And the longer they live the more progeny they have."
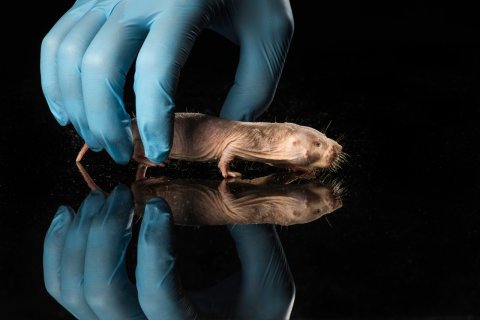
A naked mole rat is pictured at the University of Rochester January 31, 2018. J. Adam Fenster/University of Rochester
The same logic applies to humans—and it also explains why our bodies fall apart. Diseases of aging, many gerontologists now argue, are the natural consequence of the advances in modern lifespan, which now extends decades past reproductive age, and thus has not been subject to the same exquisitely efficient evolutionary sculpting that might increase our odds of surviving them. "If you put this work in an evolutionary perspective, we were not supposed to live that long," says Gerard Karsenty, who chairs the Department of Genetics and Development at Columbia University Medical Center. "Aging is an invention of mankind. No animal species has successfully cheated its own body—cheated nature—except mankind. Elephants may live for 100 years but they lived for 100 years a million years ago. Humans have outsmarted their own body."
But what does any of this have to do with Wyss-Coray and Villeda's young blood?
Can Blood from Young People Slow Aging? Silicon Valley Has Bet Billions It Will
Adam Piore
28-35 minutes
The Spanish firm Grifols set off a kerfuffle last year when it nearly doubled the going price for blood donations for a COVID-19 treatment trial. Brigham Young University in Idaho had to threaten some enterprising students with suspension to keep them from intentionally trying to contract COVID-19. The trial failed, however, and now the Barcelona-based firm is hoping to extract something far more valuable from the plasma of young volunteers: a set of microscopic molecules that could reverse the process of aging itself.
Earlier this year, Grifols closed on a $146 million-deal to buy Alkahest, a company founded by Stanford University neurologist Tony Wyss-Coray, who, along with Saul Villeda, revealed in scientific papers published in 2011 and 2014 that the blood from young mice had seemingly miraculous restorative effects on the brains of elderly mice. The discovery adds to a hot area of inquiry called geroscience that focuses on identifying beneficial elements of blood that dissipate as we age and others that accumulate and cause damage. In the last six years, Alkahest has identified more than 8,000 proteins in the blood that show potential promise as therapies. Its efforts and those of Grifols have resulted in at least six phase 2 trials completed or underway to treat a wide range of age-related diseases, including Alzheimer's and Parkinson's.
Alkahest and a growing number of other geroscience health startups signal a change in thinking about some of the most intractable diseases facing humankind. Rather than focusing solely on the etiology of individual diseases like heart disease, cancer, Alzheimer's and arthritis—or, for that matter, COVID-19—geroscientists are trying to understand how these diseases relate to the single largest risk factor of all: human aging. Their goal is to hack the process of aging itself and, in the process, delay or stave off the onset of many of the diseases most associated with growing old.
The idea that aging and illness go hand and hand is, of course, nothing new. What's new is the newfound confidence of scientists that "aging" can be measured, reverse-engineered and controlled.
Until recently, "people working on diseases did not think that aging was modifiable," says Felipe Sierra, who recently retired as director of the Division of Aging Biology, part of the National Institutes of Health. "That is actually what many medical books say: The main risk factor for cardiovascular disease is aging, but we cannot change aging so let's talk about cholesterol and obesity. For Alzheimer's, aging is the main risk factor—but let's talk about the buildup in the brain of beta-amyloid proteins. Now that is beginning to change."

Felipe Sierra, who recently retired as director of the Division of Aging Biology, part of the National Institutes of Health. Courtesy of NIH/National Institute on Aging
The result is a flood of investment money, an explosion of research into what precisely goes wrong in our bodies as we get old and the promise of clinical results down the road.
In the months before the pandemic, investors ponied up billions of dollars to fund biotechs aimed at commercializing the new science. Some biotech firms are developing drugs and infusions designed to clean up zombie-like cells and metabolic junk that accumulate with age. Others hope to infuse new vigor into flagging cellular components, such as stem cells, or spur the body into beneficial actions by adding obscure hormones or proteins, that decrease as we get older. The NIA, under its director, Richard Hodes, recently announced plans to spend about $100 million over the next five years on basic research aimed at understanding "cellular senescence."
"You have no idea how many people are interested to investing money in longevity," said Nir Barzilai, the founding director of the Institute for Aging Research at Albert Einstein College of Medicine of Yeshiva University, and the founder of a company aimed at mitochondrial health. "There are billions of dollars."
Although the vast majority of these efforts remain in preclinical development, several have recently entered FDA trials and could potentially hit the market in a few years. Some are already appearing on the gray market, raising concerns that hucksters are peddling anti-aging snake oil. Others, meanwhile, worry what might happen if these drugs actually do deliver on their promise: Will poor young people be coerced into selling their blood to elderly billionaires? Will magical anti-aging pills become the province of the Park Avenue and Hollywood rich, like facelifts, hair plugs and botox injections? Will the rest of us senile peasants be forced to watch them age backwards as we are left to wither and die?
Hacking Old Age
Getting old usually doesn't end well. Despite massive gains in human life expectancy—over the last 150 years, it has almost doubled in many developed nations, though it dipped in the past year due to the pandemic—we still haven't found a way to halt the relentless toll time takes on our bodies.

Older people practicing yoga at an assisted living facility to help stay limber; Silicon Valley is working on its own solution to the mobility issues and other problems of older age. Craig F. Walker/The Boston Globe/Getty
When we grow old, our immune systems begin to break down, creating a state of low-level inflammation that suppresses cellular regeneration and leads to those aches and pains. We may be living longer, but we are hurting. As our mitochondria cease to effectively produce energy for our cells, we spend many of those hard-won extra years taking afternoon naps. As our stem cells grow lethargic and quiescent, our muscle mass shrinks and our bones become increasingly fragile. In short, our bodies fall apart.
The first hint that hacking the biology of aging might be possible came, improbably enough, from a series of laboratory experiments on a lowly species of roundworm. In the late 1980s and early 1990s, studies of identical twins had already shown that about 30 percent of longevity in humans could be attributed to genetics. But most scientists believed the process of aging was far too complex a phenomenon to modulate simply by tweaking a couple genes or taking a pill.
Then in 1993, Cynthia Kenyon, a biologist at the University of California in San Francisco, doubled the lifespan of a worm from three weeks to six by mutating a single gene. The work suggested the possibility of drugs that could increase longevity without targeting specific diseases. It suggested that the process of aging itself might be manipulated.
At the time, a fringe group of longevity enthusiasts were, unknowingly, already trying to do what Kenyon did in roundworms through the practice of calorie restriction. This idea took off when gerontologist Roy Walford of UC Los Angeles doubled the lifespan of lab mice by restricting their caloric intake and then became a vocal advocate for the practice in humans. He penned a series of best-selling books in the 1980s and adopted a 1,600-calorie-a-day diet for the last 30 years of his life. Today, his emaciated acolytes can still be spotted at longevity conferences, meticulously measuring out their food during lunch breaks.
Emerging science now explains why there may be something to this method of bio-hacking. In humans, insulin is the hormonal signal that cues our cells to absorb sugar and convert it to energy. Along with a closely related hormone called Insulin like-Growth Factor 1 (IGF1), insulin affects a large number of other cellular processes, including the rate of cellular division, which many believe is directly related to aging. When the insulin and IGF1 in humans or analogous compounds in worms are dialed down—because we are starving, or the genes have been tweaked—a host of cellular repair mechanisms that are normally on standby kick into high-gear.
The adaptation makes sense from the perspective of evolution. Prehistoric times consisted of long periods of scarcity punctuated by precious windows of abundance. Since there was no telling how long the good times would last, our ancestors evolved the ability to grow and build fat and muscle as quickly as possible. A feast triggered the body to release insulin and IGF1, which allowed our cells to begin absorbing glucose and spurred them to pour energy into cellular reproduction and regeneration for as long as our insulin levels remain high.

Beta-amyloid plaques and tau in the brain. Courtesy of NIH/National Institute on Aging
When calories were no longer easy to come by, our bodies adjusted by lowering insulin and IGF1 levels—a cue for our cells to slow regeneration and reproduction, and instead divert energy into cellular processes most likely to facilitate our survival through cold, lean times. The human body protects cells it already has: it produces more enzymes to ensure proteins don't misfold, it ramps up the machinery designed to repair broken DNA and it breaks down cellular debris and defective cells it might otherwise ignore, scavenging for parts it can use to feed its healthier cells through the lean days. In the process, it cleans up cellular garbage that, particularly as we grow older, likely promotes low-level inflammation.
Currently there is no FDA-approved indication for drugs that target the process of aging itself. To win approval, drugs must target a specific disease. It's no coincidence that some gerontologists have chosen the popular diabetes drug metformin to serve as the "template" for a new class of FDA-approved anti-aging drugs. It works by influencing the body's sensitivity to insulin and can have an effect on the pace of metabolism and energy expenditure. If studies now underway confirm its efficacy and it wins approval, it would create an entirely new regulatory category of drugs that, rather than treating specific diseases, will work to prevent our bodies from deteriorating further once we have already developed one.
"Aging drives disease," says Albert Einstein's Barzilai, who is leading the study. "That's our point. If we stop aging, it stops driving the diseases."
A whole host of other potential anti-aging-drugs might not be far behind. Molecular biologists discovered a second important way to manipulate the aging process in the early 2000s by breeding strains of baker's yeast, fruit flies and worms to live longer. They sequenced the genomes of the hardiest strains and then worked backward to identify the specific genetic variants that seemed be associated with increased longevity. That led them to mTOR, a genetic variant that serves as the target of an anti-aging drug called rapamycin, which is now undergoing clinical trials. Among these scientific efforts is a massive longitudinal study of canine aging at the University of Washington, which has enrolled 30,000 dogs and plans to select 500 lucky pooches to participate in a rapamycin trial.
The hunt for other "control knobs" that might be targeted with drugs continues at research institutions around the world. A third approach would increase the production an enzyme called AMP-Kinase, which modulates cellular processes like growth and metabolism, based on the level of energy available for consumption. It, too, has emerged in recent years as another promising target, along with many other molecules, hormones and proteins that appear to decline with age and that play a key role in cellular repair, regeneration, protection and efficient function.
Vera Gorbunova, co-director of the Rochester Aging Research Center at the University of Rochester, and her colleagues compared 18 rodent species, including, beavers, hamsters and mice, sorted them by longevity, and looked for interesting patterns. Significantly, they found that "more robust" repair of broken DNA, the molecular-level cellular blueprints found in every one of our cells, seems to coevolve with longevity. In other words, species that live longer have a more robust ability to fix the kind of problems that inevitably occur with age. The research was recently published in the journal Cell.

Vera Gorbunova, co-director of the Rochester Aging Research Center at the University of Rochester University of Rochester
"There was a very strong correlation to how long different species live and how well they repaired DNA breaks," Gorbunova said. "We also look at why it's better. We found a protein that is very important in DNA repair and it is more active in long-lived species."
Gorbunova has done some of her most exciting work on an obscure superstar in the longevity field known as the naked mole rat, a furless, wrinkled rodent with a pair of beaver-like incisors that lives in tunnels in East Africa. The rat, sometimes referred to by its cuddly nickname, "Sand Puppy," has an unusually long-life expectancy—30 years, 10 times longer than its close cousin, the North American field mouse, which rarely makes it past three.
Gorbunova traces much of the hardiness of the mole rats to an abundance of hyaluronic acid, a major component of skin that is involved in tissue regeneration. Although mice and humans also have hyaluronic acid, the tissues of naked mole rats are "saturated with it," says Gorbunova. In addition to having strong antioxidant properties, and others that seem to attenuate the destructive consequences of the chronic, widespread inflammation that often accumulates with age, the abundance of hyaluronan also seems to prevent the growth of malignant cancer cells.
"Hyaluronan is a very nice story because we can see the possibility of translating it to humans," Gorbunova says. "We have it, but we don't have a lot of it, so I think there is room for improvement. We can find ways to increase our own levels of hyaluronan."
To Gorbunova, the differences between the mouse and naked mole rat are easily explained by evolution—their respective adaptations are geared toward increasing their chances of reproductive success. "For a mouse, the best strategy to have more progeny is to be very, very prolific very quickly because then somebody's going to eat it, and it just doesn't have a chance to live longer," she explains. "The naked mole rat lives underground and has very few predators. And they breed until very late in life. So they would evolve the mechanism to allow them to live longer and to breathe as long as possible just because they can. No one is there to eat them. And the longer they live the more progeny they have."

A naked mole rat is pictured at the University of Rochester January 31, 2018. J. Adam Fenster/University of Rochester
The same logic applies to humans—and it also explains why our bodies fall apart. Diseases of aging, many gerontologists now argue, are the natural consequence of the advances in modern lifespan, which now extends decades past reproductive age, and thus has not been subject to the same exquisitely efficient evolutionary sculpting that might increase our odds of surviving them. "If you put this work in an evolutionary perspective, we were not supposed to live that long," says Gerard Karsenty, who chairs the Department of Genetics and Development at Columbia University Medical Center. "Aging is an invention of mankind. No animal species has successfully cheated its own body—cheated nature—except mankind. Elephants may live for 100 years but they lived for 100 years a million years ago. Humans have outsmarted their own body."
But what does any of this have to do with Wyss-Coray and Villeda's young blood?